Bacitracin (Injection)
{{DrugProjectFormSinglePage |authorTag=Alberto Plate [1] |genericName=Bacitracin |aOrAn=an |drugClass=antibiotic |indicationType=treatment |indication=infants with pneumonia and empyema caused by staphylococci shown to be susceptible to the drug |hasBlackBoxWarning=Yes |adverseReactions=injection site pain |blackBoxWarningTitle=WARNING |blackBoxWarningBody=Nephrotoxicity: Bacitracin in parenteral (intramuscular) therapy may cause renal failure due to tubular and glomerular necrosis. Its use should be restricted to infants with staphylococcal pneumonia and empyema when due to organisms shown to be susceptible to bacitracin. It should be used only where adequate laboratory facilities are available and when constant supervision of the patient is possible. Renal function should be carefully determined prior to and daily during therapy. The recommended daily dose should not be exceeded and fluid intake and urinary output maintained at proper levels to avoid kidney toxicity. If renal toxicity occurs the drug should be discontinued. The concurrent use of other nephrotoxic drugs, particularly streptomycin, kanamycin, polymyxin B, polymyxin E (colistin), and neomycin should be avoided. |fdaLIADAdult=====Indications==== In accord with the statements in the "Warning Box" the use of intramuscular bacitracin is limited to the treatment of infants with pneumonia and empyema caused by staphylococci shown to be susceptible to the drug.
To reduce the development of drug-resistant bacteria and maintain the effectiveness of Bacitracin and other antibacterial drugs, Bacitracin should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
Dosage
TO BE ADMINISTERED INTRAMUSCULARLY ONLY
Infant dose: For infants under 2500 grams-900 units/kg/24 hours in 2 or 3 divided doses. For infants over 2500 grams-1,000 units/kg/24 hours, in 2 or 3 divided doses. Intramuscular injections of the solution should be given in the upper outer quadrant of the buttocks, alternating right and left and avoiding multiple injections in the same region because of the transient pain following injection.
Preparation of Solutions—Should be dissolved in sodium chloride injection containing 2 percent procaine hydrochloride. The concentration of the antibiotic in the solution should not be less than 5,000 units per mL nor more than 10,000 units per mL.
Diluents containing parabens should not be used to reconstitute bacitracin; cloudy solutions and precipitate formation have occurred.
Reconstitution of the 50,000 unit vial with 9.8 mL of diluent will result in a concentration of 5,000 units per mL.
Solutions are stable for one week when stored in a refrigerator 2° to 8°C (36° to 46°F). |offLabelAdultGuideSupport=There is limited information regarding Off-Label Guideline-Supported Use of Bacitracin in adult patients. |offLabelAdultNoGuideSupport======Pelvic Inflammatory Disease[1]=====
Pseudomembranous Enterocolitis
- Dosage: 80,000 to 100,000 units/day in divided doses for 7 to 10 days[2]
|fdaLIADPed=*Bacitracin is limited to the treatment of infants with pneumonia and empyema caused by staphylococci shown to be susceptible to the drug.To reduce the development of drug-resistant bacteria and maintain the effectiveness of Bacitracin and other antibacterial drugs, Bacitracin should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
- Dosage:
- Infants under 2500 grams-900 units/kg/24 hours in 2 or 3 divided doses.
- Infants over 2500 grams-1,000 units/kg/24 hours, in 2 or 3 divided doses.
- Dosage:
- Intramuscular injections of the solution should be given in the upper outer quadrant of the buttocks, alternating right and left and avoiding multiple injections in the same region because of the transient pain following injection.
Preparation of Solutions
- Should be dissolved in sodium chloride injection containing 2 percent procaine hydrochloride. The concentration of the antibiotic in the solution should not be less than 5,000 units per mL nor more than 10,000 units per mL. Diluents containing parabens should not be used to reconstitute bacitracin; cloudy solutions and precipitate formation have occurred. Reconstitution of the 50,000 unit vial with 9.8 mL of diluent will result in a concentration of 5,000 units per mL. Solutions are stable for one week when stored in a refrigerator 2° to 8°C (36° to 46°F).
|offLabelPedGuideSupport=There is limited information regarding Off-Label Guideline-Supported Use of Bacitracin in pediatric patients. |offLabelPedNoGuideSupport=There is limited information regarding Off-Label Non–Guideline-Supported Use of Bacitracin in pediatric patients. |contraindications=This drug is contraindicated in those individuals with a history of previous hypersensitivity or toxic reaction to it. |warnings=*Clostridium difficile associated diarrhea (CDAD) has been reported with use of nearly all antibacterial agents, including Bacitracin for Injection and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C. difficile.
- C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibiotic use. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents.
- If CDAD is suspected or confirmed, ongoing antibiotic use not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibiotic treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated.
|clinicalTrials======Nephrotoxic reactions=====
Nonspecific Symptoms
|drugInteractions=*The concurrent use of other nephrotoxic drugs, particularly streptomycin, kanamycin, polymyxin B, polymyxin E (colistin), and neomycin should be avoided. |FDAPregCat=C |monitoring=Renal function should be carefully determined prior to and daily during therapy. The recommended daily dose should not be exceeded and fluid intake and urinary output maintained at proper levels to avoid kidney toxicity. |drugBox={{Drugbox2 | Verifiedfields = changed | verifiedrevid = 457285800 | IUPAC_name = (4R)-4-[(2S)-2-({2-[(1S)-1-amino-2-methylbutyl]- 4,5-dihydro-1,3-thiazol-5-yl}formamido)-4-methylpentanamido]-4-{[(1S)- 1-{[(3S,6R,9S,12R,15S,18R,21S)- 18-(3-aminopropyl)-12-benzyl-15-(butan-2-yl)-3-(carbamoylmethyl)- 6-(carboxymethyl)-9-(1H-imidazol-5-ylmethyl)-2,5,8,11,14,17,20- heptaoxo-1,4,7,10,13,16,19-heptaazacyclopentacosan-21-yl]carbamoyl}- 2-methylbutyl]carbamoyl}butanoic acid | image = DB2 Structure.png
| tradename = Baciim | Drugs.com = Monograph | pregnancy_AU = D | pregnancy_US = C | legal_AU = S4 | legal_US = Rx-only | routes_of_administration = Topical, intramuscular
| bioavailability = | protein_bound = | metabolism = | elimination_half-life =
| CASNo_Ref =
| CAS_number_Ref =
| CAS_number = 1405-87-4
| ATC_prefix = D06
| ATC_suffix = AX05
| ATC_supplemental = J01XX10 (WHO) R02AB04 (WHO) Template:ATCvet
| PubChem = 439542
| DrugBank_Ref =
| DrugBank = DB00626
| ChemSpiderID_Ref =
| ChemSpiderID = 10481985
| UNII_Ref =
| UNII = 58H6RWO52I
| KEGG_Ref =
| KEGG = D00128
| ChEMBL_Ref =
| ChEMBL = 1200558
| C=66 | H=103 | N=17 | O=16 | S=1
| molecular_weight = 1422.69 g/mol
| smiles = O=C(O)C[C@H]3NC(=O)[C@H](Cc1cncn1)NC(=O)[C@@H](Cc2ccccc2)NC(=O)[C@@H](NC(=O)[C@@H](CCCN)NC(=O)[C@H](CCCCNC(=O)[C@H](CC(N)=O)NC3=O)NC(=O)[C@@H](NC(=O)[C@@H](CCC(=O)O)NC(=O)[C@H](CC(C)C)NC(=O)C4N=C(SC4)\nC(N)C(C)CC)C(C)CC)C(C)CC
| InChI = 1/C66H103N17O16S/c1-9-35(6)52(69)66-81-48(32-100-66)63(97)76-43(26-34(4)5)59(93)74-42(22-23-50(85)86)58(92)83-53(36(7)10-2)64(98)75-40-20-15-16-25-71-55(89)46(29-49(68)84)78-62(96)47(30-51(87)88)79-61(95)45(28-39-31-70-33-72-39)77-60(94)44(27-38-18-13-12-14-19-38)80-65(99)54(37(8)11-3)82-57(91)41(21-17-24-67)73-56(40)90/h12-14,18-19,31,33-37,40-48,52-54H,9-11,15-17,20-30,32,67,69H2,1-8H3,(H2,68,84)(H,70,72)(H,71,89)(H,73,90)(H,74,93)(H,75,98)(H,76,97)(H,77,94)(H,78,96)(H,79,95)(H,80,99)(H,82,91)(H,83,92)(H,85,86)(H,87,88)/t35?,36?,37?,40-,41+,42+,43-,44+,45-,46-,47+,48?,52?,53-,54-/m0/s1
| InChIKey = CLKOFPXJLQSYAH-NVOBBBONBV
| StdInChI_Ref =
| StdInChI = 1S/C66H103N17O16S/c1-9-35(6)52(69)66-81-48(32-100-66)63(97)76-43(26-34(4)5)59(93)74-42(22-23-50(85)86)58(92)83-53(36(7)10-2)64(98)75-40-20-15-16-25-71-55(89)46(29-49(68)84)78-62(96)47(30-51(87)88)79-61(95)45(28-39-31-70-33-72-39)77-60(94)44(27-38-18-13-12-14-19-38)80-65(99)54(37(8)11-3)82-57(91)41(21-17-24-67)73-56(40)90/h12-14,18-19,31,33-37,40-48,52-54H,9-11,15-17,20-30,32,67,69H2,1-8H3,(H2,68,84)(H,70,72)(H,71,89)(H,73,90)(H,74,93)(H,75,98)(H,76,97)(H,77,94)(H,78,96)(H,79,95)(H,80,99)(H,82,91)(H,83,92)(H,85,86)(H,87,88)/t35?,36?,37?,40-,41+,42+,43-,44+,45-,46-,47+,48?,52?,53-,54-/m0/s1
| StdInChIKey_Ref =
| StdInChIKey = CLKOFPXJLQSYAH-NVOBBBONSA-N
}}
|structure=The structural formula is:

The molecular formula is: C66H103N17O16S |PK=Bacitracin exerts pronounced antibacterial action in vitro against a variety of gram-positive and a few gram-negative organisms. However, among systemic diseases, only staphylococcal infections qualify for consideration of bacitracin therapy. Bacitracin is assayed against a standard and its activity is expressed in units, 1 mg having a potency of not less than 50 units.
Susceptibility plate testing: If the Kirby-Bauer method of disk susceptibility is used, a 10 unit bacitracin disk should give a zone of over 13 mm when tested against a bacitracin-susceptible strain of Staphylococcus aureus. Absorption of bacitracin following intramuscular injection is rapid and complete. A dose of 200 or 300 units/kg every 6 hours gives serum levels of 0.2 to 2 mcg/mL in individuals with normal renal function. The drug is excreted slowly by glomerular filtration. It is widely distributed in all body organs and is demonstrable in ascitic and pleural fluids after intramuscular
|howSupplied=

|storage=Store the unreconstituted product in a refrigerator at 2° to 8°C (36° to 46°F).
|packLabel=
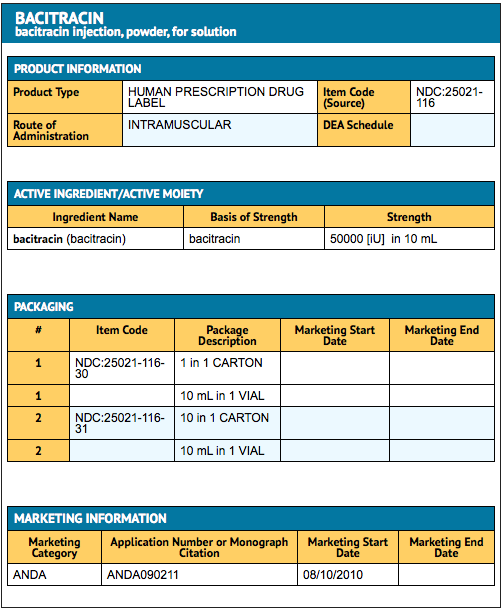
|fdaPatientInfo=Patients should be counseled that antibacterial drugs including Bacitracin should only be used to treat bacterial infections. They do not treat viral infections (e.g., the common cold). When Bacitracin is prescribed to treat a bacterial infection, patients should be told that although it is common to feel better early in the course of therapy, the medication should be taken exactly as directed.
Skipping doses or not completing the full course of therapy may:
- (1) Decrease the effectiveness of the immediate treatment
- (2) Increase the likelihood that bacteria will develop resistance and will not be treatable by Bacitracin or other antibacterial drugs in the future.
Diarrhea is a common problem caused by antibiotics which usually ends when the antibiotic is discontinued. Sometimes after starting treatment with antibiotics, patients can develop watery and bloody stools (with or without stomach cramps and fever) even as late as two or more months after having taken the last dose of the antibiotic. If this occurs, patients should contact their physician as soon as possible. |alcohol=Alcohol-Bacitracin interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication. |brandNames=*AK-Tracin
}} {{#subobject:
|Label Page=Bacitracin (Injection) |Label Name=Bacitracin Package.png
}}
- ↑ Wright VC, Hatch LA, Lanning NM (1980). "Use of a topical triple-antibiotic spray to reduce morbidity from pelvic infection after gynecologic operations". Can J Surg. 23 (4): 366–9, 372. PMID 7417900.
- ↑ Chang TW, Gorbach SL, Bartlett JG, Saginur R (1980). "Bacitracin treatment of antibiotic-associated colitis and diarrhea caused by Clostridium difficile toxin". Gastroenterology. 78 (6): 1584–6. PMID 7372074.