Arachidonic acid
|
WikiDoc Resources for Arachidonic acid |
|
Articles |
|---|
|
Most recent articles on Arachidonic acid Most cited articles on Arachidonic acid |
|
Media |
|
Powerpoint slides on Arachidonic acid |
|
Evidence Based Medicine |
|
Clinical Trials |
|
Ongoing Trials on Arachidonic acid at Clinical Trials.gov Trial results on Arachidonic acid Clinical Trials on Arachidonic acid at Google
|
|
Guidelines / Policies / Govt |
|
US National Guidelines Clearinghouse on Arachidonic acid NICE Guidance on Arachidonic acid
|
|
Books |
|
News |
|
Commentary |
|
Definitions |
|
Patient Resources / Community |
|
Patient resources on Arachidonic acid Discussion groups on Arachidonic acid Patient Handouts on Arachidonic acid Directions to Hospitals Treating Arachidonic acid Risk calculators and risk factors for Arachidonic acid
|
|
Healthcare Provider Resources |
|
Causes & Risk Factors for Arachidonic acid |
|
Continuing Medical Education (CME) |
|
International |
|
|
|
Business |
|
Experimental / Informatics |
Overview
Arachidonic acid (AA) is an omega-6 fatty acid 20:4(ω-6). It is the counterpart to the saturated arachidic acid found in peanut oil, (L. arachis – peanut.)[1]
Chemical characteristics
Chemically, arachidonic acid is a carboxylic acid with a 20-carbon chain and four cis double bonds; the first double bond is located at the sixth carbon from the omega end.
Some chemistry sources define 'arachidonic acid' to designate any of the eicosatetraenoic acids. However, almost all writings in biology, medicine and nutrition limit the term to all-cis 5,8,11,14-eicosatetraenoic acid.
Biological role
Arachidonic acid is a polyunsaturated fatty acid that is present in the phospholipids (especially phosphatidylethanolamine, phosphatidylcholine and phosphatidylinositides) of membranes of the body's cells, and is abundant in the brain.
It is also involved in cellular signaling as a second messenger.
It is the source of several other molecules with specific roles, as described in the next section.
Synthesis and cascade
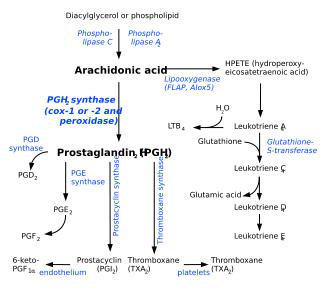
Arachidonic acid is freed from phospholipid molecule by the enzyme phospholipase A2, which cleaves off the fatty acid precursor (usually linoleic acid).
Arachidonic acid is a precursor in the production of eicosanoids:
- the enzymes cyclooxygenase and peroxidase lead to Prostaglandin H2, which in turn is used to produce the prostaglandins, prostacyclin, and thromboxanes.
- the enzyme 5-lipoxygenase leads to 5-HPETE, which in turn is used to produce the leukotrienes.
- arachidonic acid is also used in the biosynthesis of anandamide.
The production of these derivatives, and their action in the body, are collectively known as the arachidonic acid cascade; see essential fatty acid interactions for more details.
Arachidonic acid as an essential fatty acid
Arachidonic acid is one of the essential fatty acids required by most mammals. Some mammals lack the ability to—or have a very limited capacity to—convert linoleic acid into arachidonic acid, making it an essential part of their diet. Since little or no arachidonic acid is found in plants, such animals are obligate carnivores; the cat is a common example.[2][3]
- For a more detailed discussion of the term 'essential', see Essential fatty acid – What is "essential"?
References
- ↑ "Dorland's Medical Dictionary – 'A'". Retrieved 2007-01-12.
- ↑ MacDonald M, Rogers Q, Morris J (1984). "Nutrition of the domestic cat, a mammalian carnivore". Annu Rev Nutr. 4: 521–62. PMID 6380542. Retrieved 2007-02-09.
- ↑ Rivers J, Sinclair A, Crawford M (1975). "Inability of the cat to desaturate essential fatty acids". Nature. 258 (5531): 171–3. doi:10.1038/258171a0. PMID 1186900.
See also
- Polyunsaturated fat
- Polyunsaturated fatty acid
- Aspirin - inhibits cyclooxygenase enzyme to prevent the conversion of arachidonic acid to other signal molecules
External links
- Arachidonic Acid at acnp.org
- Arachidonic+Acid at the US National Library of Medicine Medical Subject Headings (MeSH)
de:Arachidonsäure it:Acido arachidonico lv:Arahidonskābe nl:Arachidonzuur sl:Arahidonska kislina fi:Arakidonihappo